Transition metal pairs on ceria-promoted, ordered mesoporous alumina as catalysts for the CO2 reforming reaction of methane†
Abstract
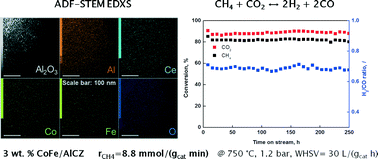
CoFe, CoW, NiFe and NiW bimetallic pairs deposited over CeO2–ZrO2 promoted ordered mesoporous γ-Al2O3 were tested in the methane dry reforming reaction. The catalysts were characterized by N2 adsorption, wide and small angle XRD, TEM, UV-vis DRS and H2-TPR techniques. Upon thermal treatment, diffusion of Ni and Co into the alumina matrix occurred, resulting in the formation of corresponding aluminate spinels. Based on UV-vis characterization, the formation of bulk Fe2O3 could be only tentatively assigned to the NiFe/AlCZ catalyst. Bulk Co3O4 is likely present in both CoW and CoFe catalysts. CoFe/AlCZ and NiFe/AlCZ catalysts achieved high and stable methane conversion rates of 8.8 and 6.0 mmol of CH4 per gcat per min, respectively. The activity of tungsten-containing catalysts (CoW/AlCZ and NiW/AlCZ) was substantially lower compared to iron-containing catalysts (2.8 and 0.8 mmol of CH4 per gcat per min, respectively) and their deactivation was likely related to sintering and oxidation of active metal clusters. A low carbon content was accumulated on the surface of spent NiFe/AlCZ, CoFe/AlCZ and NiW/AlCZ catalysts after 20 h tests (0.6–1.2 wt%), indicating the high efficiency of the redox promoter under dry reforming conditions.

 Please wait while we load your content...
Please wait while we load your content...