Catalytic hydrodeoxygenation of palmitic acid over a bifunctional Co-doped MoO2/CNTs catalyst: an insight into the promoting effect of cobalt†
Abstract
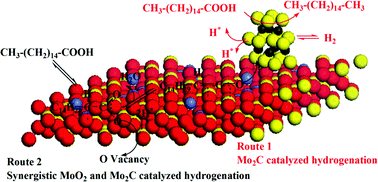
Novel Co-doped MoO2/CNTs catalysts were prepared by a wet-impregnation method and employed in catalytic hydrodeoxygenation (HDO) of palmitic acid. The obtained catalysts were systematically characterized using various techniques, namely, XRD, BET surface area, XPS, FT-IR spectroscopy of adsorbed pyridine, Raman, H2-TPD, and H2-TPR. Characterization studies revealed the doping of Co ions into the lattice of MoO2, the interaction between metal species modified the electrical properties of the catalytic active sites, and the formation of new active sites and defects. The catalytic results showed that Co ions could significantly improve catalytic performance, and the best selectivity to hexadecane reached 89.3% at an extremely low temperature of 180 °C. The increased presence of Mo2C particles, Lewis acidic sites and oxygen vacancies were all responsible for the noticeable catalytic performance of the Co doped catalyst. The mechanistic insights from this work confirmed the bifunctional role of Co-doped MoO2/CNTs catalysts for HDO of palmitic acid, which was catalyzed either solely by Mo2C or synergistically by Mo2C and MoO2. Insights into the nature of the active site would provide a useful knowledge for rational design of effective Mo-based HDO catalysts and assist future studies on more efficient catalytic conversion systems.

 Please wait while we load your content...
Please wait while we load your content...