CuO–CeO2/TiO2 catalyst for simultaneous NO reduction and Hg0 oxidation at low temperatures
Abstract
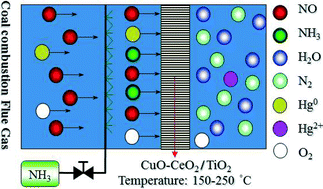
CuO–CeO2/TiO2 (CuCeTi) catalyst prepared by a sol–gel method was employed for the first time to simultaneously reduce nitrogen monoxide (NO) and oxidize elemental mercury (Hg0) in the presence of ammonia (NH3) at low flue gas temperatures. The combination of copper oxides and cerium oxides yielded apparent synergy for NO reduction and Hg0 oxidation. Over 90% of NO reduction and over 80% of Hg0 oxidation were simultaneously obtained on the CuCeTi catalyst at 200 °C to 250 °C under simulated coal combustion flue gas with a gas hourly space velocity (GHSV) of 54 000 h−1 which is more than 10 times higher than the actual GHSV in selective catalytic reduction (SCR) reactors. The well-dispersed active species and abundant chemisorbed surface oxygen on the CuCeTi catalyst were responsible for its excellent catalytic performance. Neither Hg0 vapor in flue gas nor HgO loaded on the catalyst surface inhibited NO reduction by NH3 at 200 °C. In the absence of gas phase oxygen (O2), NH3 consumed surface oxygen and hence resulted in the deactivation of Hg0 oxidation. In the presence of O2, neither NO nor NH3 individually exhibited an inhibition on Hg0 oxidation. However, Hg0 oxidation over the CuCeTi catalyst was inhibited by the co-presence of NO and HH3. Fortunately, this inhibitive effect was entirely scavenged when a lower GHSV was adopted. This study reveals the feasibility of simultaneously reducing NO and oxidizing Hg0 at low flue gas temperatures. Such knowledge is of fundamental importance in developing efficient, practical and economical NO and Hg0 control technologies for coal-fired power plants.

 Please wait while we load your content...
Please wait while we load your content...