CO oxidation activity of thermally stable Fe–Cu/CeO2 catalysts prepared by dual-mode arc-plasma process†
Abstract
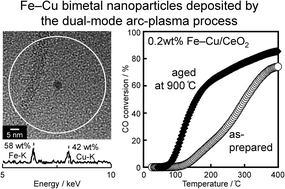
Fe–Cu bimetal nanoparticles were deposited on CeO2 powders at a low loading level (≤0.2 wt%) by the dual-mode arc-plasma (AP) process. The bimetal particles enabled the formation of Fe–Cu oxide after being thermally aged at 900 °C, which was characterised by a highly dispersed Cu+ and Fe3+ on the surface of CeO2. The CO oxidation activity was enhanced by thermal aging at 900 °C when prepared by the AP process compared to the conventional wet-impregnation method, despite the significant decrease in surface area from 170 m2 g−1 to less than 10 m2 g−1. The higher activity can be rationalised by the more efficient formation of Fe–Cu oxide, the Cu+ species of which plays the role of CO chemisorption site. The reaction rate of CO oxidation exhibited partial orders of ~0.9 and ~0.0 with respect to CO and O2, respectively. Pulsed C16O–18O2 reactions revealed that CO adsorbed on Cu+ reacted mainly with lattice oxygen, which implies that the reaction proceeds via the Mars–van Krevelen mechanism.

 Please wait while we load your content...
Please wait while we load your content...