A dinuclear iron complex with a single oxo bridge as an efficient water-oxidizing catalyst in the presence of cerium(iv) ammonium nitrate: new findings and current controversies†
Abstract
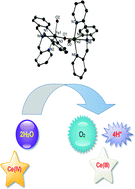
In this paper a dinuclear Fe complex, [tpa(H2O)FeOFe(H2O)tpa](ClO4)4 (tpa: tris(2-pyridylmethyl)amine) is reported as an efficient catalyst for water oxidation in the presence of cerium(IV) ammonium nitrate. The turnover frequency of this dimer is 6 times greater than the turnover frequency reported for the [Fe(OTf or Cl)2(tpa)] complex (OTf: trifluoromethanesulfonate). The role of FeO42− ions in the water oxidation reaction is also discussed.

 Please wait while we load your content...
Please wait while we load your content...