Chemoselective olefinmetathesis transformations mediated by ruthenium complexes
Abstract
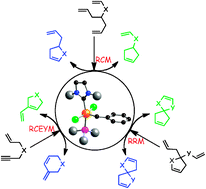
Over the past decade, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds either in ring-closing or cross-
C bonds either in ring-closing or cross-
* Corresponding authors
a
School of Chemistry, University of St Andrews, St Andrews, UK
E-mail:
snolan@st-andrews.ac.uk
b
Université Aix-Marseille, UMR CNRS 6263-Institut des Sciences Moléculaires de Marseille, Av. Escadrille Normandie Niemen,
, 13397 Marseille Cedex 20, France
E-mail:
herve.clavier@univ-cezanne.fr
Over the past decade, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds either in ring-closing or cross-
C bonds either in ring-closing or cross-

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
S. P. Nolan and H. Clavier, Chem. Soc. Rev., 2010, 39, 3305 DOI: 10.1039/B912410C
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
