Phase transition-induced changes in the Raman properties of DMSO/benzene binary systems†
Abstract
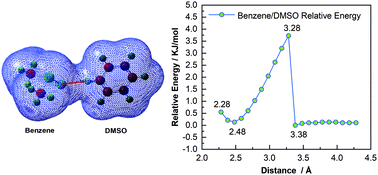
The Raman spectra of dimethylsulfoxide (DMSO)/benzene binary mixtures were studied by decreasing the temperature from 333 K to 263 K with the aim to reveal the molecular interaction properties during phase transition. The intensity of the Raman band for benzene at 992 cm−1 showed an increasing trend in the liquid and solid phases, while it exhibited a highly decreasing trend during the liquid–solid phase transition. The potential energy was calculated to study the effect of intermolecular interaction distance between DMSO and benzene on Raman intensity. The observations indicated that the blueshift of the low-frequency bands of DMSO was significantly different from the redshift of its high-frequency bands. The hydrogen bond generated between DMSO and benzene was well formed in the binary systems. This interaction inducing an enhanced hydrogen bond between the binary systems and attenuated C–H bonds led to opposite Raman shift variations with decreasing temperature. The Raman bands of DMSO at 1425 cm−1, 2899 cm−1, and 2992 cm−1 each split into two peaks after phase transition. The splitting of the Raman bands of DMSO at 1417 cm−1, 2895 cm−1, and 2982 cm−1 cropped up as the temperature dropped to the transformation point of 288 K. This is attributed to the phase transition-induced latent def.(C7) atomic vibrations corresponding to the individual methyl groups of DMSO. The implications of these analyses are expected to be helpful to understand the effect of phase transition on the Raman properties of binary solutions.
- This article is part of the themed collection: 2021 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...