Ruffling drives coproheme decarboxylation by facilitating PCET: a theoretical investigation of ChdC†
Abstract
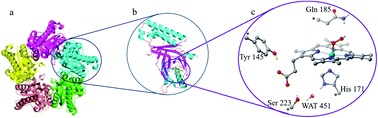
Coproheme decarboxylase (ChdC) is an essential enzyme in the coproporphyrin-dependent heme synthesis pathway, which catalyzes oxidative decarboxylation of coproheme at the positions p2 and p4 to generate heme b under the action of hydrogen peroxide. A mysterious characteristic of catalytic mechanism of ChdC is that both of the two decarboxylation sites are located remotely from the iron center of coproheme, which binds with hydrogen peroxide. By using density functional theory calculations, we have studied the coproheme decarboxylation mechanism of ChdC in detail. The calculation results show that in the first step of the catalytic reaction, H2O2 homolysis takes place synergistically with the proton coupled electron transfer process of a tyrosine (Tyr145) residing near p2 propionate. The produced reactive Tyr radical then abstracts a hydrogen atom from the β carbon of the p2 propionate side chain, which is the rate-limiting step of the whole reaction with a 19.16 kcal mol−1 energy barrier. Finally, through intramolecular electron and proton rearrangement of coproporphyrin, decarboxylation of p2 propionate is accomplished. Our study revealed that the ruffled conformation of coproheme in ChdC is an important structural factor, which facilitates the decarboxylation reaction. We also found that the hydrogen bond chain located below the coproheme ring plays a role to regulate the PCET process of Tyr145. In addition, molecular dynamics simulations discovered that Lys149 is responsible for stabilizing the harderoheme III and positioning the second decarboxylation site p4 to the catalytic Tyr145 site in the decarboxylation reaction of the p4 site.


 Please wait while we load your content...
Please wait while we load your content...