Understanding surface charge regulation in silica nanopores†
Abstract
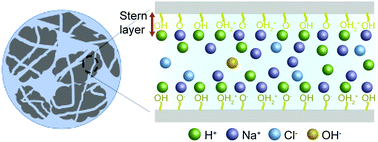
Nanoporous silica is used in a wide variety of applications, ranging from bioanalytical tools and materials for energy storage and conversion as well as separation devices. The surface charge density of nanopores is not easily measured by experiment yet plays a vital role in the performance and functioning of silica nanopores. Herein, we report a theoretical model to describe charge regulation in silica nanopores by combining the surface-reaction model and the classical density functional theory (CDFT). The theoretical predictions provide quantitative insights into the effects of pH, electrolyte concentration, and pore size on the surface charge density and electric double layer structure. With a fixed pore size, the surface charge density increases with both pH and the bulk salt concentration similar to that for an open surface. At fixed pH and salt concentration, the surface charge density rises with the pore size until it reaches the bulk asymptotic value when the surface interactions become negligible. At high pH, the surface charge density is mainly determined by the ratio of the Debye screening length to the pore size (λD/D).


 Please wait while we load your content...
Please wait while we load your content...
