Fluorescence measurements of peroxynitrite/peroxynitrous acid in cold air plasma treated aqueous solutions
Abstract
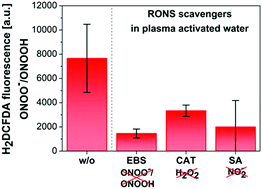
Qualitative detection of peroxynitrite/peroxynitrous acid (ONOO−/ONOOH) as one of the key bactericidal agents produced in cold air plasma activated aqueous solutions is presented. We examined the use of the 2,7-dichlorodihydrofluorescein diacetate (H2DCFDA) fluorescent dye to detect ONOO−/ONOOH in plasma activated non-buffered water (PAW) or buffered solution (PAPB) generated by DC-driven self-pulsed transient spark discharge at atmospheric pressure in ambient air. The diagnostic selectivity of H2DCFDA to reactive oxygen and nitrogen species (RONS) typical of plasma activated aqueous solutions was examined by using various scavengers of RONS. This cross-reactivity study showed the highest sensitivity of the H2DCFDA dye to ONOO−/ONOOH. However, besides ONOO−/ONOOH, H2DCFDA also exhibited sensitivity to hypochlorite anions/hypochlorous acid (OCl−/HOCl), showing that for a selective study it is important to have an idea about the possible constituents in the studied solutions. The sensitivity of H2DCFDA to other RONS even in much higher concentrations was negligible. The presence of nitrites (NO2−) and hydrogen peroxide (H2O2) in PAW led predominantly to the production of peroxynitrous acid with a strong fluorescence response of H2DCFDA in PAW. Plasma treatment of buffered solutions led to the weak response of H2DCFDA. The fluorescence induced in PAW decreased after scavenging individual reactants, namely NO2− and H2O2, as well as by scavenging the product of the peroxynitrite forming reaction, proving that the fluorescence response of H2DCFDA is primarily due to the formation of ONOO−/ONOOH. A chemical kinetics analysis of post-discharge processes and the pseudo-second order reaction between H2O2 and NO2− confirms formation of peroxynitrous acid in PAW with a rate in the order of tens of nM per second. The post-discharge evolution of the ONOOH formation rate was clearly correlated with the parallel detection of ONOO−/ONOOH by fluorescence spectroscopy using the H2DCFDA dye.


 Please wait while we load your content...
Please wait while we load your content...