Partition of electronic excitation energies: the IQA/EOM-CCSD method†
Abstract
Different developments in chemistry and emerging technologies have generated a renewed interest in the properties of molecular excited states. We present herein the partition of black-box, size-consistent equation-of-motion coupled cluster singles and doubles (EOM-CCSD) excitation energies within the framework of the interacting quantum atoms (IQA) formalism. We denote this method as IQA/EOM-CCSD. We illustrate this approach by considering small molecules used often in the study of excited states. This investigation shows how the combination of IQA and EOM-CCSD may provide valuable insights into the molecular changes induced by electron excitation via the real space distribution of the energy of an absorbed photon in a molecular system. Our results reveal (i) the most energetically deformed atomic basins and (ii) the most affected covalent and non-covalent interactions within a molecule due to a given photoexcitation. In other words, this kind of analysis provides insights into the spatial energetic redistribution accompanying an electronic excitation, with interesting foreseeable applications in the rational design of photoexcitations with tailored chemical effects. Altogether, we expect that the IQA/EOM-CCSD excitation energy partition will prove useful in the understanding of systems and processes of interest in photophysics and photochemistry.
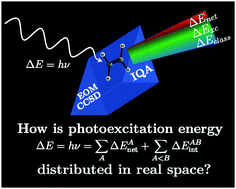
- This article is part of the themed collections: Celebrating Latin American Talent in Chemistry, Celebrating recent chemical science in Mexico and 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...