Determination of band edges and their influences on photocatalytic reduction of nitrobenzene by bulk and exfoliated g-C3N4†
Abstract
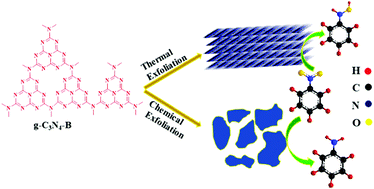
Thermally and chemically exfoliated metal-free semiconducting g-C3N4 are synthesized from bulk g-C3N4. Thorough characterization of the synthesized materials is performed with the help of XRD, FTIR, FE-SEM, PL, surface area analysis and DRS to probe differences in structural, morphological and optical properties between thermally and chemically exfoliated g-C3N4. The synthesized materials are exposed to light for photocatalytic reduction of nitrobenzene. The complete reduction reaction mechanism and product selectivity over the synthesized catalysts are studied in this report. The rate of reduction of nitrobenzene is found to be higher with thermally exfoliated g-C3N4, and the selectivity of aniline is found to be higher in the case of chemical exfoliated g-C3N4. The differences in the reactivity are explained in terms of structure, surface morphologies and band edge positions.


 Please wait while we load your content...
Please wait while we load your content...