Mixed-dimensional 2D/3D heterojunctions between MoS2 and Si(100)†
Abstract
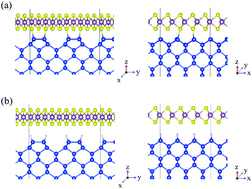
For utilization of two-dimensional (2D) materials as electronic devices, their mixed-dimensional heterostructures with three-dimensional (3D) materials are receiving much attention. In this study, we have investigated the atomic and electronic structures of the 2D/3D heterojunction between MoS2 and Si(100) using density functional theory calculations; especially, we focus on the contact behavior dependence on the interfacial structures of heterojunctions by considering two types of surface termination of Si(100) surfaces. Calculations show that MoS2 and clean Si(100) form an almost n-type ohmic contact with a very small Schottky barrier height (SBH) due to strong covalent bonds between them, and that the contact between MoS2 and H-covered Si(100) makes a p–n heterojunction with weak van der Waals interactions. Such a difference in contact behaviors can be explained by different electric dipole formation at the heterojunction interfaces. Overall, it is concluded that contact properties can be varied depending on the interfacial structures of 2D(MoS2)/3D(Si) semiconductor heterojunctions.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...