Dynamics changes of CRISPR-Cas9 systems induced by high fidelity mutations†
Abstract
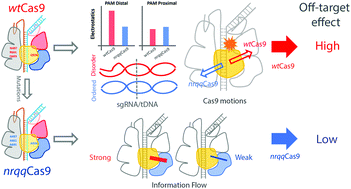
CRISPR-Cas9, a powerful genome editing tool, has widely been applied in biological fields. Since the discovery of CRISPR-Cas9 as an adaptive immune system, it has been gradually modified to perform precise genome editing in eukaryotic cells by creating double-strand breaks. Although it is robust and efficient, the current CRISPR-Cas9 system faces a major flaw: off-target effects, which are not well understood. Several Cas9 mutants show significant improvement, with very low off-target effects; however, they also show relatively lower cleavage efficiency for on-target sequences. In this study, the dynamics of wild-type Cas9 from Streptococcus pyogenes and a high fidelity Cas9 mutant have been explored using molecular dynamics simulations. It was found that the mutations cause decreased electrostatic interactions between Cas9 and the R-loop. Consequently, the flexibility of the tDNA/sgRNA heteroduplex is decreased, which may explain the lower tolerance of mismatches in the heteroduplex region. The mutations also affect the protein dynamics and the correlation networks among Cas9 domains. In mutant Cas9, weakened communications between two catalytic domains as well as a slight opening of the conformation induced by the mutations account for the lower on-target cleavage efficiency and probably the lower off-target efficiency as well. These findings will facilitate more precise Cas9 engineering in future.


 Please wait while we load your content...
Please wait while we load your content...