Influence of membrane lipid composition on the structure and activity of γ-secretase†
Abstract
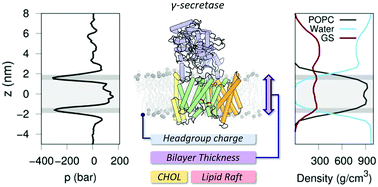
γ-Secretase (GS) is a multi-subunit membrane-embedded aspartyl protease that cleaves more than 80 integral membrane proteins, including the amyloid precursor protein (APP) to produce the amyloid-β (Aβ) peptide. Oligomerization and aggregation of the 42-amino acid length Aβ isoform in the brain has been associated with the development and progression of Alzheimer's disease (AD). Based on recent experimental structural studies and using multiscale computational modeling approaches, the conformational states and protein–membrane interactions of the GS complex embedded in six homogeneous and six heterogeneous lipid bilayers were characterized. In order to identify potential lipid and cholesterol binding sites, GS regions with high lipid/cholesterol occupancy values were analyzed using atomistic and coarse-grained simulations. Long lipid residence times were observed to be correlated with a large number of hydrogen bonds between the charged headgroups and key GS amino acids. This observation provides a plausible explanation for the inhibition of GS by charged lipids observed in previous experimental studies. Computed lateral pressure profiles suggest that higher transmembrane pressures favor active state conformations of the catalytic subunit. A probable mechanism for the regulation of the local stress response in cholesterol-rich multicomponent lipid bilayers is identified. Finally, it is demonstrated that interactions between the nicastrin extracellular domain and lipid headgroups leads to a compact structural conformation of the GS complex. Overall, this study provides valuable insight into the effect of bilayer lipid composition on the GS structural ensemble and its function.
- This article is part of the themed collections: Celebrating recent chemical science in Mexico and 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...