An extended excited-state intramolecular proton transfer (ESIPT) emitter for random lasing applications†
Abstract
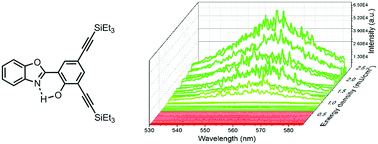
An original molecular fluorophore displaying single keto (K*) excited-state intramolecular proton transfer (ESIPT) emission is presented in this article. Substitution at the 3 and 5 positions of the phenol ring of a 2-(2′-hydroxyphenylbenzoxazole) (HBO) dye by triethylsilyl-ethynyl fragments leads to a drastic enhancement of fluorescence in the solution-state as compared to unsubstituted analogues. This intense fluorescence emission is also retained in a protic solvent like ethanol and in the solid-state as embedded in a potassium bromide pellet or as 1% doped in a poly(methyl methacrylate) (PMMA) film. The experimental optical properties were confirmed by ab initio calculations. Random lasing (RL) studies performed on this ESIPT emitter show the presence of stimulated emission occurring above the threshold level of pumping energy density (ρth ≈ 300 μJ cm−2) in the PMMA matrix. To the best of our knowledge, this study constitutes the first observation of RL based on ESIPT fluorescence.


 Please wait while we load your content...
Please wait while we load your content...