Binding indirect greenhouse gases OCS and CS2 by nitrogen heterocyclic carbenes (NHCs)†
Abstract
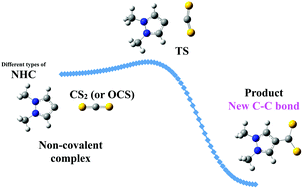
Carbon disulfide (CS2) and carbonyl sulfide (OCS) are indirect greenhouse gases that can be effectively trapped by classical, abnormal and remote nitrogen heterocyclic carbenes (NHCs), according to high level ab initio calculations. The process is described through a reaction profile involving two minima, a non-covalent complex and a covalently bound product, connected by a single transition state. Both CS2 and OCS react towards NHCs in a similar way, forming a new C–C bond and leading to very stable products with feasible barriers in many cases, although they vary significantly depending on the NHC structure. The barriers are larger than those reported for CO2, oscillating from barrierless processes up to a maximum of 57.9 kJ mol−1, whereas the products are more stabilized than those incorporating CO2. The lowest barriers for the CS2 + NHC reactions correspond to the largest C–C distances in the products, unlike the CO2 case. Remarkably, the most favored reactions, which are those involving a remote NHC, do not exhibit the highest interaction energies at the TS, but low distortion energy values of the OCS/CS2 moieties. The decomposition of the interaction energy allowed to confirmed that in fact the remote carbene is the less favored one in terms of the electrostatic and exchange terms. Substitution in CO2 of O by a more polarizable atom such as S have a great influence on the balance between the steric plus orbital interaction and the kinetic energy terms, thus making the products between NHCs and OCS/CS2 more stable. Both OCS and CS2 become better charge acceptors than CO2 on going from the starting complexes to the products.


 Please wait while we load your content...
Please wait while we load your content...