How does the total charge and isomerism influence the Ru–NO ammine complexes?†
Abstract
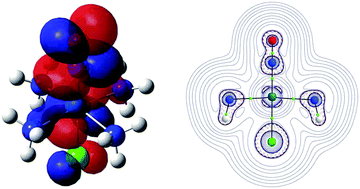
Nitric oxide plays an important role in several physiological processes. This study investigates model ruthenium ammine coordination compounds to control NO bioavailability: cis-[RuCl(NO)(NH3)4]+ (1+), cis-[RuCl(NO)(NH3)4]2+ (12+), cis-[RuCl(NO)(NH3)4]3+ (13+), trans-[RuCl(NO)(NH3)4]+ (2+), trans-[RuCl(NO)(NH3)4]2+ (22+), trans-[RuCl(NO)(NH3)4]3+ (23+), [Ru(NO)(NH3)5]+ (3+), [Ru(NO)(NH3)5]2+ (32+), and [Ru(NO)(NH3)5]3+ (33+). We employed natural population analysis (NPA) atomic charges (qNPA) and the LUMO to identify the main reduction sites in the complexes 1, 2 and 3. For example, in the transformations 12+ → 1+, 22+ → 2+, and 33+ → 32+, the main reduction site was a NO π* orbital, which accounted for the lower electron density of the Ru–NO bond critical point (BCP) in 1+, 2+, and 32+ than 12+, 22+, and 33+, respectively, as shown by the quantum theory of atoms in molecules (QTAIM). The QTAIM method indicated that the electron density was larger in Ru–NO BCP due to the Cl negative cis- and trans-influence in 12+ and 22+, respectively, as compared with the NH3 influence in 33+. Compared to trans-Cl–Ru–NO in 22+, the interacting quantum atoms method demonstrated that cis-Cl–Ru–NO in 12+ displayed (i) a larger repulsive electrostatic energy, which agreed with qNPA, and (ii) a less negative exchange–correlation energy between Ru and the NO nitrogen atom, which agreed with topological analyses performed by the QTAIM method. Thus, the combination of topological and energy decomposition analyses allowed the mechanism behind the Ru–NO bond to be revealed regarding the influence of the total charge and the relative position of the ligands.


 Please wait while we load your content...
Please wait while we load your content...