Using porphyrin–amino acid pairs to model the electrochemistry of heme proteins: experimental and theoretical investigations†
Abstract
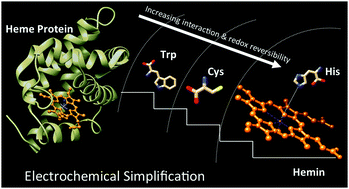
Quasi reversibility in electrochemical cycling between different oxidation states of iron is an often seen characteristic of iron containing heme proteins that bind dioxygen. Surprisingly, the system becomes fully reversible in the bare iron–porphyrin complex: hemin. This leads to the speculation that the polypeptide bulk (globin) around the iron–porphyrin active site in these heme proteins is probably responsible for the electrochemical quasi reversibility. To understand the effect of such polypeptide bulk on iron–porphyrin, we study the interaction of specific amino acids with the hemin center in solution. We choose three representative amino acids–histidine (a well-known iron coordinator in bio-inorganic systems), tryptophan (a well-known fluoroprobe for proteins), and cysteine (a redox-active organic molecule). The interactions of these amino acids with hemin are studied using electrochemistry, spectroscopy, and density functional theory. The results indicate that among these three, the interaction of histidine with the iron center is strongest. Further, histidine maintains the electrochemical reversibility of iron. On the other hand, tryptophan and cysteine interact weakly with the iron center but disturb the electrochemical reversibility by contributing their own redox active processes to the system. Put together, this study attempts to understand the molecular interactions that can control electrochemical reversibility in heme proteins. The results obtained here from the three representative amino acids can be scaled up to build a heme–amino acid interaction database that may predict the electrochemical properties of any protein with a defined polypeptide sequence.


 Please wait while we load your content...
Please wait while we load your content...