Graphene induced electrical percolation enables more efficient charge transport at a hybrid organic semiconductor/graphene interface†
Abstract
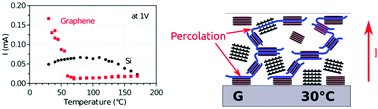
Self-assembly of semiconducting polymer chains during crystallization from a liquid or melt dictates to a large degree the electronic properties of the resulting solid film. However, it is still unclear how charge transport pathways are created during crystallization. Here, we performed complementary in situ electrical measurements and synchrotron grazing incidence X-ray diffraction (GIXD), during slow cooling from the melt of highly regio-regular poly(3-hexylthiophene) (P3HT) films deposited on both graphene and on silicon. Two different charge transport mechanisms were identified, and were correlated to the difference in crystallites' orientations and overall amount of crystallites in the films on each surface as molecular self-assembly proceeded. On silicon, a weak charge transport was enabled as soon as the first edge-on lamellae formed, and further increased with the higher amount of crystallites (predominantly edge-on and randomly oriented lamellae) during cooling. On graphene however, the current remained low until a minimum amount of crystallites was reached, at which point interconnection of conducting units (face-on, randomly oriented lamellae and tie-chains) formed percolated conducting pathways across the film. This lead to a sudden rapid increase in current by ≈10 fold, and strongly enhanced charge transport, despite a much lower amount of crystallites than on silicon.


 Please wait while we load your content...
Please wait while we load your content...