Lyophobicity may not be the main driving force for long chain surfactants from the bulk phase to the interface
Abstract
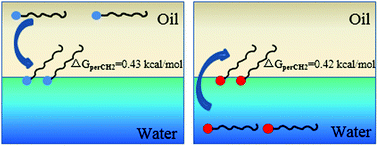
According to the Traube rule, a surfactant with a longer alkane chain is more hydrophobic so its tendency to be driven from a polar solvent to a less polar interface is higher. In this work, we revisited this topic by studying the adsorption of quaternary ammonium salts and carboxylic acids with various alkane chain lengths at the hexadecane–water interface. The adsorption free energies of the surfactants at this oil–water interface from the polar (aqueous solution) or nonpolar phase (hexadecane) were estimated from second harmonic generation measurements. The variation of the free energies per methylene group in the bulk phase, at the oil–water interface and at the air–water interface revealed that there are different interactions between the alkane chains of the surfactants in different environments. The chain–chain interaction at the hexadecane–water interface is lower than that at the air–water interface. The driving force for the alkane chains to adsorb at the oil–water interface from the oil phase is close to that from the aqueous phase. This observation reveals that the chain–chain interaction rather than the lyophobicity of the solute with respect to the solvent is the main contributor to the adsorption free energy. This is the first experimental comparison of the free energies of the alkane chains in oil, in water, at the air–water interface and at the oil–water interface. These results provide information for studying the interactions of hydrophobic species in different environments. This work also provides a method for estimating the solvation energy of some head groups in surfactants.


 Please wait while we load your content...
Please wait while we load your content...