Modeling of the oxygen reduction reaction for dense LSM thin films
Abstract
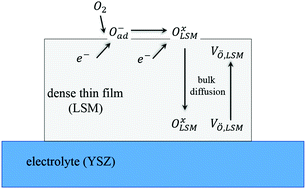
In the present study, the oxygen reduction reaction mechanism is investigated using numerical methods on a dense thin (La1−xSrx)yMnO3±δ film deposited on a YSZ substrate. This 1-D continuum model consists of defect chemistry and elementary oxygen reduction reaction steps coupled via reaction rates. The defect chemistry model contains eight species including cation vacancies on the A- and B-sites. The oxygen vacancy is calculated by solving species transportation equations in multiphysics simulations. Due to the simple geometry of a dense thin film, the oxygen reduction reaction was reduced to three elementary steps: surface adsorption and dissociation, incorporation on the surface, and charge transfer across the LSM/YSZ interface. The numerical simulations allow for calculation of the temperature- and oxygen partial pressure-dependent properties of LSM. The parameters of the model are calibrated with experimental impedance data for various oxygen partial pressures at different temperatures. The results indicate that surface adsorption and dissociation is the rate-determining step in the ORR of LSM thin films. With the fine-tuned parameters, further quantitative analysis is performed. The activation energy of the oxygen exchange reaction and the dependence of oxygen non-stoichiometry on oxygen partial pressure are also calculated and verified using the literature results.


 Please wait while we load your content...
Please wait while we load your content...