Activation of CO2 by supported Cu clusters†
Abstract
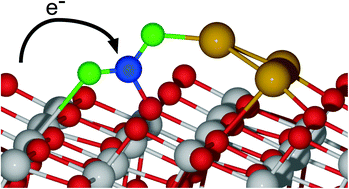
Catalytic reduction of carbon dioxide to useful chemicals is a potent way to mitigate this greenhouse gas, but the challenge lies in finding active reduction catalysts. Using density functional theory we studied CO2 activation over TiO2-supported Cu clusters of size 1–4 atoms. The linear to bent transformation of CO2 is necessary for activation, and we found that all the clusters stabilized bent CO2, along with a significant gain of electrons on the CO2 (indicative of activation). On all the TiO2 supported Cu clusters, the interfacial sites were found to stabilize the bent CO2 adsorption, where the active site of adsorption on Cu dimer, trimer and tetramer was on the Cu atom farthest away from the TiO2 surface. Particularly, the Cu dimer stabilized bent CO2 very strongly, although this species was found to be unstable on the surface. A synthesis technique that could stabilize the Cu dimer could therefore lead to a very active catalyst. Furthermore we found (using vibrational and charge analysis) that the active sites for the CO2 activation predominantly had 0 and +1 oxidation states; the oxidation state of Cu is known to directly affect CO2 reduction activity. Our study shows TiO2-supported small Cu clusters can be active catalysts for CO2 reduction and also provides further motivation for theoretical and experimental studies of metal clusters.


 Please wait while we load your content...
Please wait while we load your content...