Study of Ce–Cu mixed oxide catalysts by in situ electrical conductivity measurements
Abstract
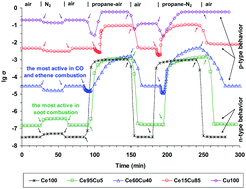
Three Ce–Cu mixed oxides, namely Ce0.95Cu0.05, Ce0.6Cu0.4 and Ce0.15Cu0.85, along with pure CeO2 and CuO were characterized by in situ electrical conductivity measurements. Their electrical conductivity was studied as a function of temperature and oxygen partial pressure, and was followed with time during successive exposure to air, nitrogen and different gaseous mixtures containing propane as a VOC model molecule, under conditions close to those of their catalytic applications. CeO2 and CuO appeared to be n-type and p-type semiconductors, respectively, while the semiconducting behavior of the Ce–Cu mixed oxides depended on the oxide composition. The semiconductive and redox properties of the samples were correlated with their catalytic behavior in CO oxidation, ethene total oxidation and soot combustion.


 Please wait while we load your content...
Please wait while we load your content...