Probing the triplet correlation function in liquid water by experiments and molecular simulations†
Abstract
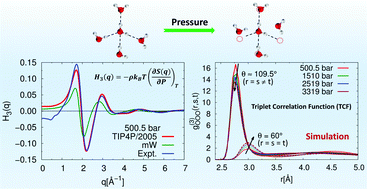
Despite very significant developments in scattering experiments like X-ray and neutron diffraction, it has been challenging to elucidate the nature of tetrahedral molecular configurations in liquid water. A key question is whether the pair correlation functions, which can be obtained from scattering experiments, are sufficient to describe the tetrahedral ordering of water molecules. In our previous study (Dhabal et al., J. Chem. Phys., 2014, 141, 174504), using data-sets generated from reverse Monte Carlo and molecular dynamics simulations, we showed that the triplet correlation functions contain important information on the tetrahedrality of water in the liquid state. In the present study, X-ray scattering experiments and molecular dynamics (MD) simulations are used to link the isothermal pressure derivative of the structure factor with the triplet correlation functions for water. Triplet functions are determined for water up to 3.3 kbar at 298 K to display the effect of pressure on the water structure. The results suggest that triplet functions (![[H with combining tilde]](https://www.rsc.org/images/entities/i_char_0048_0303.gif) (q)) obtained using a rigid-body TIP4P/2005 water model are consistent with the experimental results. The triplet functions obtained in experiment as well as in simulations evince that in the case of tetrahedral liquids, exertion of higher pressure leads to a better agreement with the Kirkwood superposition approximation (KSA). We further validate this observation using the triplet correlation functions (g(3)(r,s,t)) calculated directly from simulation trajectory, revealing that both
(q)) obtained using a rigid-body TIP4P/2005 water model are consistent with the experimental results. The triplet functions obtained in experiment as well as in simulations evince that in the case of tetrahedral liquids, exertion of higher pressure leads to a better agreement with the Kirkwood superposition approximation (KSA). We further validate this observation using the triplet correlation functions (g(3)(r,s,t)) calculated directly from simulation trajectory, revealing that both ![[H with combining tilde]](https://www.rsc.org/images/entities/i_char_0048_0303.gif) (q) in q-space and g(3)(r,s,t) in real-space contain similar information on the tetrahedrality of liquids. This study demonstrates that the structure factor, even though it has only pair correlation information of the liquid structure, can shed light on three-body correlations in liquid water through its isothermal pressure derivative term.
(q) in q-space and g(3)(r,s,t) in real-space contain similar information on the tetrahedrality of liquids. This study demonstrates that the structure factor, even though it has only pair correlation information of the liquid structure, can shed light on three-body correlations in liquid water through its isothermal pressure derivative term.

 Please wait while we load your content...
Please wait while we load your content...