Two different critical regimes enclosed in the Bean–Rodbell model and their implications for the field dependence and universal scaling of the magnetocaloric effect
Abstract
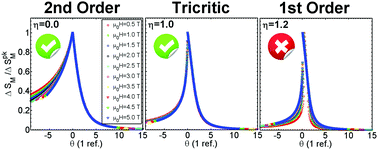
In the last few years power laws and universal scaling have been extensively used to study the field dependence of the magnitudes involved in the magnetocaloric effect of materials. They are key tools which allow us to compare the performing properties of different materials regardless of their nature, processing or experimental conditions during measurements. It was proved that power laws and universal scaling are a direct consequence of critical phenomena in the neighborhood of phase transitions. However, there remains some controversy about the reliability of these procedures. In this work we use the well-known Bean–Rodbell model to confirm that these features are unmistakably related to the critical behavior of the continuous phase transitions. In this specific model, universal scaling occurs either at a purely mean field second order transition or at a tricritical point. Finally, we analyze in detail if the universal scaling is compatible with materials at the tricitical point, making a comprehensive comparison with available experimental data from the literature. We conclude that it is really difficult to know with full certainty if a sample really is in the tricritical regime.


 Please wait while we load your content...
Please wait while we load your content...