A comparative AFM study of the interfacial nanostructure in imidazolium or pyrrolidinium ionic liquid electrolytes for zinc electrochemical systems
Abstract
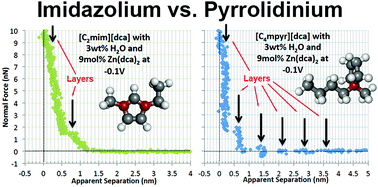
The electrochemical systems containing zinc dicyanamide salt (Zn(dca)2) in both 1-ethyl-3-methylimidazolium dicyanamide ([C2mim][dca]) and N-butyl-N-methylpyrrolidinium dicyanamide ([C4mpyr][dca]) ionic liquids (ILs) have been studied by atomic force microscopy (AFM) on a highly oriented pyrolytic graphite (HOPG) surface under different conditions and applied potentials. The results reveal the following: (1) interfacial layers exist in both ILs, even after the addition of 3 wt% water and 9 mol% Zn(dca)2 salt. (2) The number of layers is different for the different ILs, with the [C2mim][dca]-based samples exhibiting a much more limited interfacial structure compared to the [C4mpyr][dca] at almost all of the tested conditions. (3) For the [C4mpyr][dca]-based samples, without added zinc salt, the number of detected interfacial layers increases with negative potential. With added zinc, the [C4mpyr][dca] sample shows about the same number of layers independent of the applied potentials, namely between 5–7. Likewise, for the [C2mim][dca] samples, with the zinc added the sample shows the same number of layers at the applied potentials, but for this system only 1–2 layers are detected. And (4) the addition of Zn(dca)2 into the [C2mim][dca] IL does not cause a large change in the interfacial ordering, whereas the addition of the same salt into the [C4mpyr][dca] samples is marked by a stark increase in both the number and the consistency of the perceived interfacial layers. These results are significant because they show a marked difference in the interfacial nanostructure between two zinc-based electrochemical systems that were previously shown to have distinctly different electrochemical behaviour, despite their chemical similarity.


 Please wait while we load your content...
Please wait while we load your content...