Pyrite surface environment drives molecular adsorption: cystine on pyrite(100) investigated by X-ray photoemission spectroscopy and low energy electron diffraction
Abstract
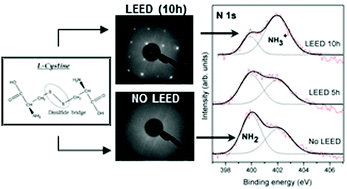
We have demonstrated that the annealing process for cleaning pyrite surfaces is a critical parameter in promoting ordering on the surface and driving surface reactivity. Furthermore, we describe a spectroscopic surface characterization of the presence or absence of the surface ordering, as indicated by the Low Energy Electron Diffraction (LEED) pattern, as a function of the surface annealing process. Complementary X-ray photoemission spectroscopy (XPS) results provide evidence that longer annealing processes of over 3 hours repair the sulfur vacancies in the pyrite, making FeS species partially disappear in favor of FeS2 species. These features play an important role in molecular adsorption. We show that in the case of the cystine amino acid on the (100) pyrite surface, the substrate structure is responsible for the chemical adsorption form. The presence of an ordered structure on the surface, as indicated by the LEED pattern, favors the cystine NH3+ chemical form, whereas the absence of the surface ordering promotes cystine NH2 adsorption due to the sulfur-deficient surface. The cystine molecule could then act by changing its chemical functionalities to compensate for the iron surface coordination. The chemical molecular adsorption form can be selected by the surface annealing conditions, implying that environmental conditions could drive molecular adsorption on mineral surfaces. These findings are relevant in several surface processes, and they could play a possible role in prebiotic chemistry surface reactions and iron–sulfur scenarios.
- This article is part of the themed collection: Prebiotic chemistry and the molecular origins of life

 Please wait while we load your content...
Please wait while we load your content...