Insight into the pseudo π-hole interactions in the M3H6⋯(NCF)n (M = C, Si, Ge, Sn, Pb; n = 1, 2, 3) complexes
Abstract
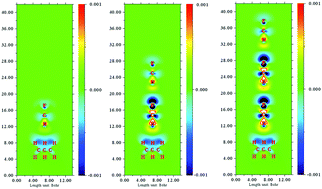
For cyclopropane and its derivatives M3H6 (M = C, Si, Ge, Sn, Pb), “pseudo π-hole” regions above and below the M–M–M three-membered ring have been discovered, and pseudo π-hole interactions between M3H6 and F–CN have been designed and investigated by MP2/aug-cc-pVTZ and MP2/aug-cc-pVTZ-pp calculations. To investigate the enhancing effects of F⋯N halogen bonds on the pseudo π-hole interactions, the termolecular and tetramolecular complexes M3H6⋯(NCF)n (n = 2, 3) were constructed. Energy decomposition analysis shows that the dispersion term contributes the most among the three attractive components in the C3H6⋯(NCF)n (n = 1, 2, 3) complexes while in the Si3H6⋯(NCF)n and Ge3H6⋯(NCF)n complexes, the electrostatic term has the largest contribution. The electrostatic and polarization energies have more effect than the dispersion energy for the enhancement of the F⋯N halogen bond on the pseudo π-hole interactions. With the increase in the number of NCF units from 1 to 3, the VS,min values outside the nitrogen atom of NCF become increasingly negative, the electric field of the lone pair of nitrogen becomes greater and causes a further increase of electron density outside the nitrogen atom and a further decrease of electron density outside the pseudo π-hole region, resulting in a stronger pseudo π-hole interaction.

 Please wait while we load your content...
Please wait while we load your content...