Contribution of convection and diffusion to the cascade reaction kinetics of β-galactosidase/glucose oxidase confined in a microchannel†
Abstract
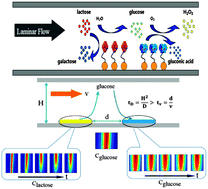
The spatial positioning of enzymes and mass transport play crucial roles in the functionality and efficiency of enzyme cascade reactions. To fully understand the mass transport regulating kinetics of enzyme cascade reactions, we investigated the contribution of convective and diffusive transports to a cascade reaction of β-galactosidase (β-Gal)/glucose oxidase (GOx) confined in a microchannel. β-Gal and GOx are assembled on two separated gold films patterned in a polydimethylsiloxane (PDMS) microchannel with a controllable distance from 50 to 100 μm. Experimental results demonstrated that the reaction yield increases with decreasing distance between two enzymes and increasing substrate flow rate. Together with the simulation results, we extracted individual reaction kinetics of the enzyme cascade reaction and found that the reaction rate catalyzed by β-Gal occurred much faster than by GOx, and thus, the β-Gal catalytic reaction showed diffusion controll, whereas the GOx catalytic reaction showed kinetic controll. Since the decrease in the enzymes distance shortens the transport length of intermediate glucose to GOx, the amount of glucose reaching GOx will be increased in the unit time, and in turn, the enzyme cascade reaction yield will be increased with decreasing the gap distance. This phenomenon is similar to the intermediates pool of tricarboxylic acid (TCA) cycle in the metabolic system. This study promotes the understanding of the metabolic/signal transduction processes and active transport in biological systems and promises to design high performance biosensors and biofuel cells systems.

 Please wait while we load your content...
Please wait while we load your content...