Electrochemistry of layered GaSe and GeS: applications to ORR, OER and HER†
Abstract
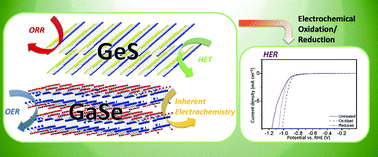
Though many studies examined the properties of the class of IIIA–VIA and IVA–VIA layered materials, few have delved into the electrochemical aspect of such materials. In light of the burgeoning interest in layered structures towards various electrocatalytic applications, we endeavored to study the inherent electrochemical properties of representative layered materials of this class, GaSe and GeS, and their impact towards electrochemical sensing of redox probes as well as catalysis of oxygen reduction, oxygen evolution and hydrogen evolution reactions. In contrast to the typical sandwich structure of MoS2 layered materials, GeS is isoelectronic to black phosphorus with the same structure; GaSe is a layered material consisting of GaSe sheets bonded in the sequence Se–Ga–Ga–Se. We characterized GaSe and GeS by employing scanning electron microscopy, X-ray diffraction, Raman spectroscopy and X-ray photoelectron spectroscopy complemented by electronic structure calculations. It was found that the encompassing surface oxide layers on GaSe and GeS greatly influenced their electrochemical properties, especially their electrocatalytic capabilities towards hydrogen evolution reaction. These findings provide fresh insight into the electrochemical properties of these IIIA–VIA and IVA–VIA layered structures which enables development for future applications.
- This article is part of the themed collection: 2016 most accessed PCCP articles

 Please wait while we load your content...
Please wait while we load your content...