Kinetics of the unimolecular reaction of CH2OO and the bimolecular reactions with the water monomer, acetaldehyde and acetone under atmospheric conditions†
Abstract
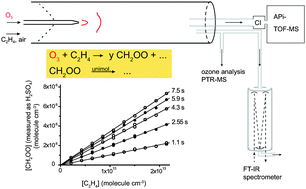
Stabilized Criegee Intermediates (sCIs) have been identified as oxidants of atmospheric trace gases such as SO2, NO2, carboxylic acids or carbonyls. The atmospheric sCI concentrations, and accordingly their importance for trace gas oxidation, are controlled by the rate of the most important loss processes, very likely the unimolecular reactions and the reaction with water vapour (monomer and dimer) ubiquitously present at high concentrations in the troposphere. In this study, the rate coefficients of the unimolecular reaction of the simplest sCI, formaldehyde oxide, CH2OO, and its bimolecular reaction with the water monomer have been experimentally determined at T = (297 ± 1) K and at atmospheric pressure by using a free-jet flow system. CH2OO was produced by the reaction of ozone with C2H4, and CH2OO concentrations were probed indirectly by detecting H2SO4 after titration with SO2. Time-resolved experiments yield a rate coefficient of the unimolecular reaction of k(uni) = (0.19 ± 0.07) s−1, a value that is supported by quantum-chemical and statistical rate theory calculations as well as by additional measurements performed under CH2OO steady-state conditions. A rate coefficient of k(CH2OO+H2O) = (3.2 ± 1.2) × 10−16 cm3 molecule−1 s−1 has been determined for sufficiently low H2O concentrations (<1015 molecule cm−3) that allow separation from the CH2OO reaction with the water dimer. In order to evaluate the accuracy of the experimental approach, the rate coefficients of the reactions with acetaldehyde and acetone were reinvestigated. The obtained rate coefficients k(CH2OO+acetald) = (1.7 ± 0.5) × 10−12 and k(CH2OO+acetone) = (3.4 ± 0.9) × 10−13 cm3 molecule−1 s−1 are in good agreement with literature data.

 Please wait while we load your content...
Please wait while we load your content...