Effects of stereoelectronic interactions on the relativistic spin–orbit and paramagnetic components of the 13C NMR shielding tensors of dihaloethenes†
Abstract
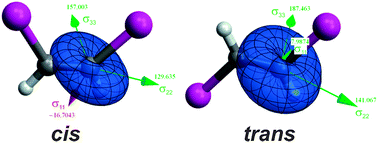
In this study, stereoelectronic interactions were considered to explain the experimental difference in the magnitude of the known heavy-atom effect on the 13C NMR chemical shifts in cis- and trans-1,2-dihaloethene isomers (halo = F, Cl, Br or I). The experimental values were compared to the calculated values with various DFT functionals using both the nonrelativistic approach (NR) and the relativistic approximations SR-ZORA (SR) and SO-ZORA (SO). NBO and NLMO contributions to the 13C NMR shielding tensors were determined to assess which stereoelectronic interactions have a more important effect on the shielding tensor in each principal axis system (PAS) coordinate. These analyses associated with the orbital rotation model and the HOMO–LUMO energy gap enable rationalization of trends between cis and trans isomers from fluorine to iodine derivatives. Both paramagnetic and SO shielding terms were responsible for the observed trends. It was possible to conclude that the steric interactions between the two iodine atoms and the hyperconjugative interactions involving the halogen lone pairs (LP(X)) and πC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C*, σC
C*, σC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C* and σC–X* antibonding orbitals are responsible for the lower 13C NMR shielding for the cis isomers of the bromine and the iodine compounds than that of the trans isomers.
C* and σC–X* antibonding orbitals are responsible for the lower 13C NMR shielding for the cis isomers of the bromine and the iodine compounds than that of the trans isomers.

 Please wait while we load your content...
Please wait while we load your content...