Role of lattice defects in catalytic activities of graphene clusters for fuel cells
Abstract
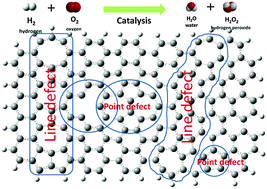
Defects are common but important in graphene, which could significantly tailor the electronic structures and physical and chemical properties. In this study, the density functional theory (DFT) method was applied to study the electronic structure and catalytic properties of graphene clusters containing various point and line defects. The electron transfer processes in oxygen reduction reaction (ORR) on perfect and defective graphene clusters in fuel cells was simulated, and the free energy and reaction energy barrier of the elementary reactions were calculated to determine the reaction pathways. It was found that the graphene cluster with the point defect having pentagon rings at the zigzag edge, or line defects (grain boundaries) consisting of pentagon–pentagon–octagon or pentagon–heptagon chains also at the edges, shows the electrocatalytic capability for ORR. Four-electron and two-electron transfer processes could occur simultaneously on graphene clusters with certain types of defects. The energy barriers of the reactions are comparable to that of platinum(111). The catalytic active sites were determined on the defective graphene.

 Please wait while we load your content...
Please wait while we load your content...