A key discovery at the TiO2/dye/electrolyte interface: slow local charge compensation and a reversible electric field
Abstract
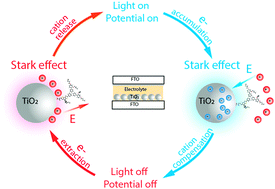
Dye-sensitized mesoporous TiO2 films have been widely applied in energy and environmental science related research fields. The interaction between accumulated electrons inside TiO2 and cations in the surrounding electrolyte at the TiO2/dye/electrolyte interface is, however, still poorly understood. This interaction is undoubtedly important for both device performance and fundamental understanding. In the present study, Stark effects of an organic dye, LEG4, adsorbed on TiO2 were well characterized and used as a probe to monitor the local electric field at the TiO2/dye/electrolyte interface. By using time-resolved photo- and potential-induced absorption techniques, we found evidence for a slow (t > 0.1 s) local charge compensation mechanism, which follows electron accumulation inside the mesoporous TiO2. This slow local compensation was attributed to the penetration of cations from the electrolyte into the adsorbed dye layer, leading to a more localized charge compensation of the electrons inside TiO2. Importantly, when the electrons inside TiO2 were extracted, a remarkable reversal of the surface electric field was observed for the first time, which is attributed to the penetrated and/or adsorbed cations now being charge compensated by anions in the bulk electrolyte. A cation electrosorption model is developed to account for the overall process. These findings give new insights into the mesoporous TiO2/dye/electrolyte interface and the electron–cation interaction mechanism. Electrosorbed cations are proposed to act as electrostatic trap states for electrons in the mesoporous TiO2 electrode.

 Please wait while we load your content...
Please wait while we load your content...