Elucidating the conformational energetics of glucose and cellobiose in ionic liquids†
Abstract
A major challenge for the utilization of lignocellulosic feedstocks for liquid fuels and other value added chemicals has been the recalcitrant nature of crystalline cellulose to various hydrolysis techniques. Ionic liquids (ILs) are considered to be a promising solvent for the dissolution and conversion of cellulose to simple sugars, which has the potential to facilitate the unlocking of biomass as a supplement and/or replacement for petroleum as a feedstock. Recent studies have revealed that the orientation of the hydroxymethyl group, described via the ω dihedral, and the glycosidic bond, described via the φ–ψ dihedrals, are significantly modified in the presence of ILs. In this study, we explore the energetics driving the orientational preference of the ω dihedral and the φ–ψ dihedrals for glucose and cellobiose in water and three imidazolium based ILs. It is found that interactions between the cation and the ring oxygen in glucose directly impact the conformational preference of the ω dihedral shifting the distribution towards the gauche–trans (GT) conformation and creating an increasingly unfavorable gauche–gauche (GG) conformation with increasing tail length. This discovery modifies the current hypothesis that intramolecular hydrogen bonding is responsible for the shift in the ω dihedral distribution and illuminates the importance of the cation's character. In addition, it is found that the IL's interaction with the glycosidic bond results in the modification of the observed φ–ψ dihedrals, which may have implications for hydrolysis in the presence of ILs. The molecular level information gained from this study identifies the favorable IL–sugar interactions that need to be exploited in order to enhance the utilization of lignocellulosic biomass as a ubiquitous feedstock.
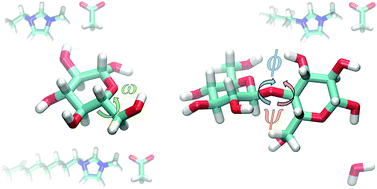

 Please wait while we load your content...
Please wait while we load your content...