Non-covalent interactions in water electrolysis: influence on the activity of Pt(111) and iridium oxide catalysts in acidic media
Abstract
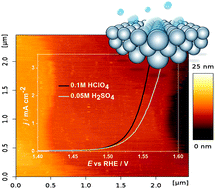
Electrolyte components, which are typically not considered to be directly involved in catalytic processes at solid–liquid electrified interfaces, often demonstrate a significant or even drastic influence on the activity, stability and selectivity of electrocatalysts. While there has been certain progress in the understanding of these electrolyte effects, lack of experimental data for various important systems frequently complicates the rational design of new active materials. Modern proton-exchange membrane (PEM) electrolyzers utilize Pt- and Ir-based electrocatalysts, which are among the very few materials that are both active and stable under the extreme conditions of water splitting. We use model Pt(111) and Ir-oxide films grown on Ir(111) electrodes and explore the effect of alkali metal cations and sulfate-anions on the hydrogen evolution and the oxygen evolution reactions in acidic media. We demonstrate that sulfate anions decrease the activity of Ir-oxide towards the oxygen evolution reaction while Rb+ drastically promotes hydrogen evolution reaction at the Pt(111) electrodes as compared to the reference HClO4 electrolytes. Issues related to the activity benchmarking for these catalysts are discussed.
- This article is part of the themed collection: Bunsentagung 2015: Solvation Science

 Please wait while we load your content...
Please wait while we load your content...