o-Benzyne fragmentation and isomerization pathways: a CASPT2 study†
Abstract
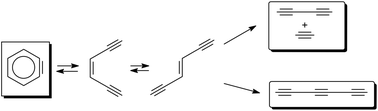
The mechanisms of the fragmentation and isomerization pathways of o-benzyne were studied at the multi-configurational second-order perturbative level [CAS(12,12)-PT2]. The direct fragmentation of o-benzyne to C2H2 + C4H2 follows two mechanisms: a concerted mechanism and a stepwise mechanism. Although the concerted mechanism is characterized by a single closed-shell transition structure, the stepwise pathway is more complex and structures with a strong diradical character are seen. A third diradicaloid fragmentation pathway of o-benzyne yields C6H2 as the final product. As an alternative to fragmentation, o-benzyne can also undergo rearrangement to its meta and para isomers and to the open chain cis and trans isomers of hexa-3-en-1,6-diyne (HED). These easily fragment to C2H2 + C4H2 or C6H2. Kinetic modelling at several different temperatures between 800 and 3000 K predicted that the thermal decomposition of o-benzyne should yield C2H2, C4H2 and C6H2 as the main products. Small amounts of the HED isomers accumulated at temperatures <1200 K, but they rapidly decompose at higher temperatures. Between 1000 and 1400 K, C2H2 + C4H2 are formed exclusively from the decomposition of trans-HED. At temperatures >1400 K, C2H2 + C4H2 also form from the direct fragmentation of o-benzyne. The formation of C2H2 + C4H2 prevails up to 1600 K but above this temperature the formation of C6H2 prevails. At temperatures >2400 K, the direct fragmentation of o-benzyne again leads to the formation of C2H2 + C4H2. The formation of hydrogen atoms is also explained by our proposed mechanisms.

 Please wait while we load your content...
Please wait while we load your content...