Is HO3− multiple-minimum and floppy? Covalent to van der Waals isomerization and bond rupture of a peculiar anion
Abstract
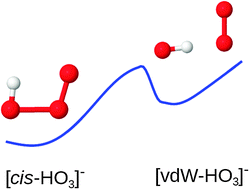
The singlet ground-state of the HO3− anion is studied with high level single- and multireference methods, and the scheme termed complete-active-space-dynamical-correlation, which has been previously used to study the neutral HO3 radical. It is found to have a planar cis isomeric structure with a long intermediate O–O bond (≈1.75 Å), as is now consensual in the literature. It also has a pyramidal-type branch-isomer, but its minimum lies ≈33 kcal mol−1 above the cis minimum. Interestingly, another isomer is predicted here, with a planar geometry that can be even more stable than cis-HO3− at some levels of theory. It shows a hydrogen-bond (van der Waals) type structure, with an intermediate O–O bond of ≈2.59 Å. All such minima lie on the lowest adiabatic potential energy surface, with the two lowest planar ones (cis and vdW) connected by a saddle point whose structure, also planar, is unveiled. All these lie on the first third of the optimum path for bond-rupture in [HO–OO]−, which is predicted to yield ground state HO plus O2−, an asymptote lying 30 kcal mol−1 above the cis-HO3− minimum. Unprecedented in the literature on the key title anion, such features should bear strong implications for its preparation, spectroscopy, and role in chemistry.

 Please wait while we load your content...
Please wait while we load your content...