The use of UV/ozone-treated MoS2 nanosheets for extended air stability in organic photovoltaic cells
Abstract
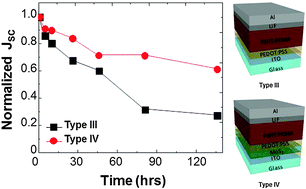
MoS2 nanosheets obtained through a simple sonication exfoliation method are employed as a hole-extraction layer (HEL) to improve the efficiency and air stability of organic photovoltaic cells (OPVs). The reduction in the wavenumber difference, appearance of a UV-vis peak, and atomic force microscopy images indicate that MoS2 nanosheets are formed through the sonication method. The OPVs with MoS2 layers show a degraded performance with a power conversion efficiency (PCE) of 1.08%, which is lower than that of OPVs without HEL (1.84%). After performing the UV/ozone (UVO) treatment of the MoS2 surface for 15 min, the PCE value increases to 2.44%. Synchrotron radiation photoelectron spectroscopy data show that the work function of MoS2 increases from 4.6 to 4.9 eV upon UVO treatment, suggesting that the increase in the PCE value is caused by the bandgap alignment. Upon inserting poly(3,4-ethylenedioxythiophene):poly(styrene sulfonate) (PEDOT:PSS) between MoS2 and the active layer, the PCE value of the OPV increases to 2.81%, which is comparable with that of the device employing only PEDOT:PSS. Furthermore, the stability of the OPVs is improved significantly when MoS2/PEDOT:PSS layers are used as the HEL. Therefore, it is considered that the use of UVO-treated MoS2 may improve the stability of OPV cells without degrading the device performance.

 Please wait while we load your content...
Please wait while we load your content...