Liquid state dynamic nuclear polarization of ethanol at 3.4 T (95 GHz)
Abstract
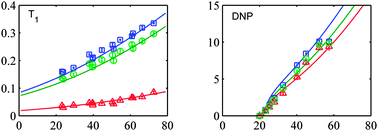
Dynamic Nuclear Polarization (DNP) in the liquid state has become the focus of attention to improve the NMR sensitivity of mass limited samples. The Overhauser model predicts a fast reduction in DNP enhancement at high magnetic fields where the electron Larmor frequency exceeds the typical inverse correlation time of the magnetic interaction between an unpaired electron spin of a radical and proton spins of the solvent molecules. The Overhauser hard sphere model is able to predict quantitatively the DNP enhancement for water TEMPOL solutions. The increase in temperature due to dielectric heating of the sample acts to reduce the correlation times and allows a substantial Overhauser DNP. In this paper we extend the work done on water towards other small molecules, such as ethanol. Experimentally we observe a similar enhancement for all three proton groups in the ethanol molecule. The classical interpretation of the low field Overhauser experiments on ethanol invokes a very fast anisotropic rotation of the hydrogen bonded TEMPOL–ethanol complex to explain the fast relaxation of the OH proton. Here we will discuss W-band relaxation and DNP enhancement within this classical model. Although the description can be made quantitative, the invoked parameters do not seem to be realistic. We will propose an alternative model based on the dynamic interaction both in free collision and due to modulation of the hydrogen bond length of the complex.

 Please wait while we load your content...
Please wait while we load your content...