Self-ignited high temperature synthesis and enhanced super-exchange interactions of Ho3+–Mn2+–Fe3+–O2− ferromagnetic nanoparticles
Abstract
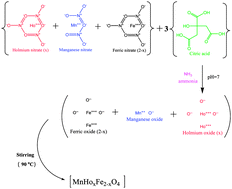
The present work is focused on the effect of Fe3+ replacement by rare earth-Ho3+ ions and their influence on the properties of MnFe2O4 ferrite. The Ho3+ substituted MnFe2O4 ferrite samples with chemical formula MnHoxFe2−xO4 were synthesized where substitution concentration of Ho3+ was 0.0, 0.05, 0.1 and 0.15. The samples were synthesized by the self-ignited sol–gel method using the nitrates of the respective elements. Powder X-ray diffraction, transmission electron microscopy, infrared spectroscopy, vibrating sample magnetometer (VSM) and electrical measurements were employed to characterize the structural, magnetic and electrical properties of these ferrite nanoparticles. The cations distribution between the tetrahedral (A-site) and octahedral sites (B-site) has been estimated by XRD analysis. It is found that substitution of Ho3+ ions favorably influenced the magnetic and electrical properties. Magnetic measurements were carried out at 77 and 300 K. Saturation magnetization and coercivity increased from 54.57 to 71.6 emu g−1 and 172 to 766 Oe, respectively, with increasing the Ho3+ substitution. The change in magnetic properties may be explained with the increase of A–O–B (FeA3+–O2−–HoB3+) super exchange interactions and the anisotropy constant. The electrical properties show that the pure sample has lower resistivity with respect to any Ho3+ doped one. The conduction mechanism is used to interpret electrical measurements. Results of the presently investigated samples with enhanced saturation magnetization, coercivity and remanence ratio indicate that the Ho3+ doped MnFe2O4 nanoparticles can be a useful candidate for the application in high density recording media.

 Please wait while we load your content...
Please wait while we load your content...