Effects of Zn2+ and Pb2+ dopants on the activity of Ga2O3-based photocatalysts for water splitting†
Abstract
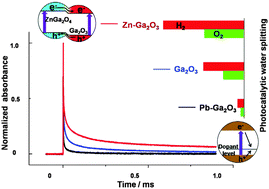
Zn-doped and Pb-doped β-Ga2O3-based photocatalysts were prepared by an impregnation method. The photocatalyst based on the Zn-doped β-Ga2O3 shows a greatly enhanced activity in water splitting while the Pb-doped β-Ga2O3 one shows a dramatic decrease in activity. The effects of Zn2+ and Pb2+ dopants on the activity of Ga2O3-based photocatalysts for water splitting were investigated by HRTEM, XPS and time-resolved IR spectroscopy. A ZnGa2O4–β-Ga2O3 heterojunction is formed in the surface region of the Zn-doped β-Ga2O3 and a slower decay of photogenerated electrons is observed. The ZnGa2O4–β-Ga2O3 heterojunction exhibits type-II band alignment and facilitates charge separation, thus leading to an enhanced photocatalytic activity for water splitting. Unlike Zn2+ ions, Pb2+ ions are coordinated by oxygen atoms to form polyhedra as dopants, resulting in distorted surface structure and fast decay of photogenerated electrons of β-Ga2O3. These results suggest that the Pb dopants act as charge recombination centers expediting the recombination of photogenerated electrons and holes, thus decreasing the photocatalytic activity.

 Please wait while we load your content...
Please wait while we load your content...