The effect of fluorine substitution on chiral recognition: interplay of CH⋯π, OH⋯π and CH⋯F interactions in gas-phase complexes of 1-aryl-1-ethanol with butan-2-ol†
Abstract
The molecular diastereomeric complexes between R-1-phenyl-1-ethanol, S-1-(4-fluorophenyl)ethanol and S-1-(2-fluorophenyl)ethanol and R and S-butan-2-ol, isolated under molecular beam conditions in the gas phase, have been investigated by mass-selective resonant two-photon ionization (R2PI) and infrared depleted R2PI (IR-R2PI). The comparison of the three systems allowed us to highlight the significance of specific intermolecular interactions in the chiral discrimination process. The interpretation of the results is based on theoretical predictions mainly at the D-B3LYP/6-31++G** level of theory. The homo and heterochiral complexes are endowed with fine differences in intermolecular interactions, namely strong OH⋯O, and weaker CH⋯π, OH⋯π, CH⋯F as well as repulsive interactions. The presence of a fluorine atom in the para position of the aromatic ring does not influence the overall geometry of the complex whilst it affects the electron density in the π system and the strength of CH⋯π and OH⋯π interactions. The role and the importance of CH⋯F intermolecular interactions are evident in the complexes with fluorine substitution in the ortho position. While the ortho hetero complex is structurally analogous to the hetero para and non-fluorinated structures, butan-2-ol in the ortho homo adduct adopts a different conformation in order to establish a CH⋯F intermolecular interaction.
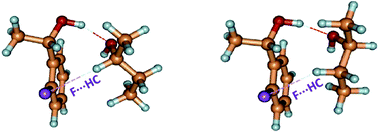

 Please wait while we load your content...
Please wait while we load your content...