Structural and electrochemical characterization of carbon supported Pt–Pr catalysts for direct ethanol fuel cells prepared using a modified formic acid method in a CO atmosphere†
Abstract
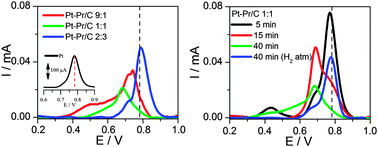
Pt–Pr/C electrocatalysts were prepared using a modified formic acid method, and their activity for carbon monoxide and ethanol oxidation was compared to Pt/C. No appreciable alloy formation was detected by XRD analysis. By TEM measurements it was found that Pt particle size increases with an increasing Pr content in the catalysts and with decreasing metal precursor addition time. XPS measurements indicated Pt segregation on the catalyst surface and the presence of Pr2O3 and PrO2 oxides. The addition of Pr increased the electro-catalytic activity of Pt for both CO and CH3CH2OH oxidation. The enhanced activity of Pt–Pr/C catalysts was ascribed to both an electronic effect, caused by the presence of Pr2O3, and the bi-functional mechanism, caused by the presence of PrO2.

 Please wait while we load your content...
Please wait while we load your content...