Ultrafast dynamics of the ns (n = 3,4) and 3d Rydberg states of O2
Abstract
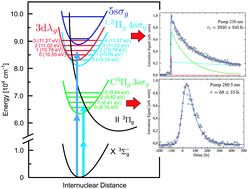
The ultrafast dynamics of the lowest optically accessible Rydberg states of molecular oxygen (O2) has been studied by time resolved transient ionization. The process was triggered by the absorption of two pump

 Please wait while we load your content...
Please wait while we load your content...