The electronic structure of pyracene: a spectroscopic and computational study†
Abstract
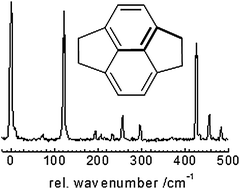
We report a synthetic, spectroscopic and computational study of the polycyclic aromatic molecule pyracene, which contains aliphatic five-membered rings annealed to a
- This article is part of the themed collection: Bunsentagung 2013: 'Theory meets Spectroscopy'

 Please wait while we load your content...
Please wait while we load your content...