Multi-structural variational transition state theory: kinetics of the 1,5-hydrogen shift isomerization of the 1-butoxyl radical including all structures and torsional anharmonicity†
Abstract
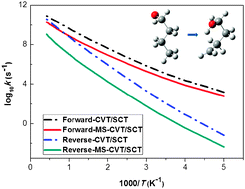
We investigate the statistical thermodynamics and kinetics of the 1,5-hydrogen shift isomerization reaction of the 1-butoxyl radical and its reverse isomerization. The partition functions and thermodynamic functions (entropy, enthalpy, heat capacity, and Gibbs free energy) are calculated using the multi-structural torsional (MS-T) anharmonicity method including all structures for three species (reactant, product, and transition state) involved in the reaction. The calculated thermodynamic quantities have been compared to those estimated by the empirical group additivity (GA) method. The kinetics of the unimolecular isomerization reaction was investigated using multi-structural canonical variational transition state theory (MS-CVT) including both multiple-structure and torsional (MS-T) anharmonicity effects. In these calculations, multidimensional tunneling (MT) probabilities were evaluated by the small-curvature tunneling (SCT) approximation and compared to results obtained with the zero-curvature tunneling (ZCT) approximation. The high-pressure-limit rate constants for both the forward and reverse reactions are reported as calculated by MS-CVT/MT, where MT can be ZCT or SCT. Comparison with the rate constants obtained by the single-structural harmonic oscillator (SS-HO) approximation shows the importance of anharmonicity in the rate constants of these reactions, and the effect of multi-structural anharmonicity is found to be very large. Whereas the tunneling effect increases the rate constants, the MS-T anharmonicity decreases them at all temperatures. The two effects counteract each other at temperatures 385 K and 264 K for forward and reverse reactions, respectively, and tunneling dominates at lower temperatures while MS-T anharmonicity has a larger effect at higher temperatures. The multi-structural torsional anharmonicity effect reduces the final reverse reaction rate constants by a much larger factor than it does to the forward ones as a result of the existence of more low-energy structures of the product 4-hydroxy-1-butyl radical than the reactant 1-butoxyl radical. As a consequence there is also a very large effect on the equilibrium constant. The neglect of multi-structural anharmonicity will lead to large errors in the estimation of reverse reaction rate constants.

 Please wait while we load your content...
Please wait while we load your content...