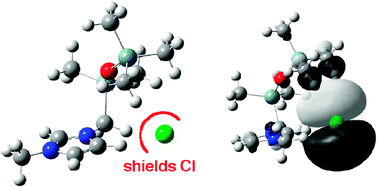
In this paper we use ab initio theoretical methods in combination with experimental studies to investigate ion-pairs of the ionic liquid (IL) 1-methyl-3-pentamethyldisiloxymethylimidazolium chloride [(SiOSi)C1C1im]Cl, in order to deepen our understanding of the effects of functionalisation on an IL. In addition, we focus on the effect of the siloxy group on the viscosity. We establish that the ion-pairing energies of [(SiOSi)C1C1im]Cl are similar to those of 1-butyl-3-methylimidazolium chloride [C4C1im]Cl, because the anion interacts primarily with the imidazolium ring. A large range of ion-pair structural configurations is possible with different anion positions and chain orientations, contributing to a significant entropy. A H-bonded network forms, however the siloxy chain can shield the Cl− or key C–H sites thus introducing defects. Despite a significant increase in mass relative to [C4C1im]+, the combined barriers to rotation within the substituent chain are substantially reduced in [(SiOSi)C1C1im]+, this is primarily due to the flexibility of the siloxane linkage, and free rotation of the Si–Me methyl groups. The most important effect is a coupling of rotational motions within the chain which leads to dynamic inter-conversion of cation conformers, and facilitates movement of the anion around the cation, these will contribute to enhanced transport properties and a reduced viscosity. In addition, a longer charge arm is expected to enhance rotational and rotational-translational coupling in electric fields. Thus, for [(SiOSi)C1C1im]Cl ion-pair association is very similar to that of [C4C1im]Cl, but “dynamic” properties relating to torsional motion, a dynamic H-bonded network, and cation response to an external electric field are enhanced.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...