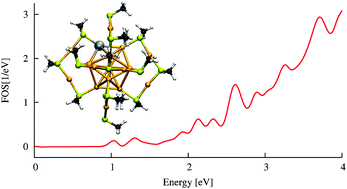
Structure, electronic properties, optical absorption and charging properties of methylthiolate-protected bimetal PdAu24(SR)18z (R = Me) clusters with various charge states (−3 ≤z≤ +3) are investigated by using density functional theory. The results are compared to properties of the well-understood singly anionic pure gold complex Au25(SR)18(−1) [J. Akola, M. Walter, H. Häkkinen and H. Grönbeck, J. Am. Chem. Soc., 2008, 130, 3756]. The atomic structure of this all-gold complex can be written in a “divide-and-protect” way [H. Häkkinen, M. Walter and H. Grönbeck, J. Phys. Chem. B, 2006, 110, 9927] as Au13[Au2(SR)3]6(−1) where 6 v-shaped Au2(SR)3 ligands protect the close-to-icosahedral Au13 core and where eight delocalized metal electrons, derived from Au(6s) electrons, comprise a stable closed-shell 1S21P6“superatom” configuration in the core. We show that the di-anion PdAu24(SR)18(−2) is a corresponding eight-electron closed-shell species whereas the clusters PdAu24(SR)18z, −1 ≤z≤ +3, have holes in the 1P HOMO manifold. This indicates that the doping Pd atom remains close to the zero-valent 4d105s0 configuration and does not contribute electrons to the delocalized electron density in the gold core. Structural optimization shows that the all-gold “divide-and-protect” structure motif is robust with respect to replacing the Au by Pd at the center of the core, at the surface of the core or in one of the protecting Au2(SR)3 ligands. However, optical absorption and the HOMO–LUMO and electrochemical gaps depend sensitively on the site of the doping Pd atom, which may turn out be useful for assigning the structure of PdAu24(SR)18 from experimental data.

 Please wait while we load your content...
Please wait while we load your content...